How a thin membrane becomes a battlefield — and what science is doing to win the war?
Every 18 seconds, somewhere in the world, a person develops bacterial meningitis. Half of survivors carry permanent damage — hearing loss, amputated limbs, or cognitive impairment — for the rest of their lives. Here is the paradox that haunts neurologists: the immune system’s attempt to protect the brain is often what destroys it. Understanding meningitis means understanding one of biology’s most tragic cases of friendly fire, and the race to stop it before the body becomes its own worst enemy.
Q1: What exactly is meningitis and why can it become life-threatening so fast?
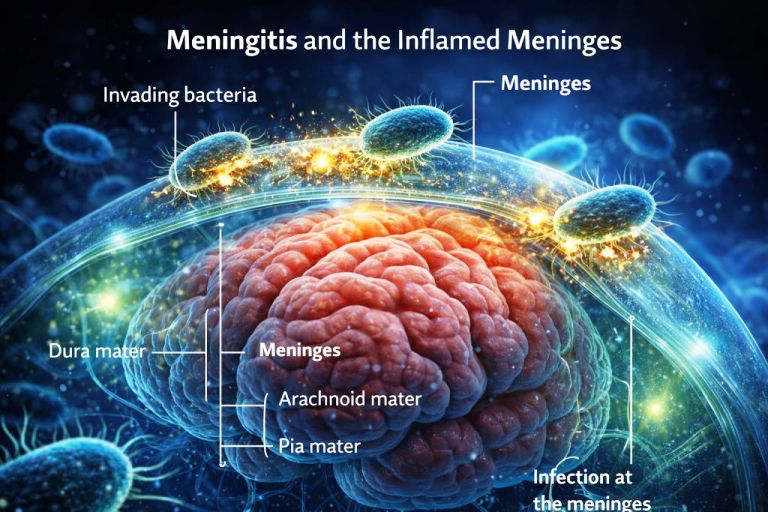
Meningitis is an inflammation of the meninges—the membranes surrounding the brain and spinal cord—usually triggered by infection.
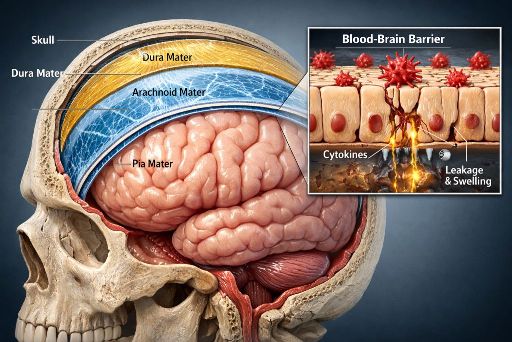
The meninges include three layers: dura mater, arachnoid mater, and pia mater. They surround cerebrospinal fluid (CSF), which cushions neural tissue and transports nutrients. When bacteria or viruses invade this space, immune cells release cytokines—chemical signals that summon additional defenders.
Research led by neurologist Allan Tunkel at Brown University shows that these cytokines increase blood vessel permeability. Fluid leaks into brain tissue and pressure inside the skull rises. Because the skull cannot expand, neurons quickly lose oxygen and glucose.
Think of it like firefighters flooding a burning building faster than drains can remove the water. The rescue effort itself causes structural damage. Understanding which microbes ignite this response leads to the next question.
Q2: What microbes actually cause meningitis?
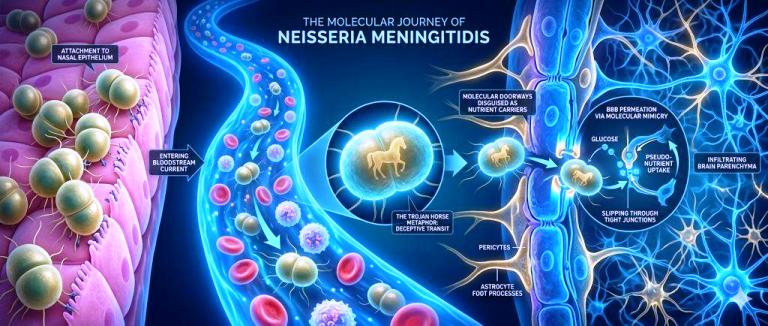
Several pathogens can infect the meninges, but bacteria and viruses account for most cases.The most dangerous form is bacterial meningitis, commonly caused by *Neisseria meningitidis* and *Streptococcus pneumoniae*. These microbes often live harmlessly in the nose or throat. Under certain conditions they enter the bloodstream and cross the blood-brain barrier—a tightly sealed layer of endothelial cells that normally blocks pathogens.
Microbiologists at the U.S. National Institutes of Health have shown that some bacteria bind to receptors on these cells and effectively trick the barrier into transporting them into the brain. Once inside CSF, bacteria multiply rapidly because immune surveillance there is limited.
Viruses such as enteroviruses cause milder but far more common cases. Yet infection alone does not determine outcomes. The speed of detection often decides survival.
Q3: Why is meningitis in the news in the United States right now?
Recent clusters of meningococcal disease have prompted public-health warnings and renewed vaccination campaigns.
The Centers for Disease Control and Prevention reported that U.S. meningococcal infections rose above pre-pandemic levels after 2022, with several outbreaks linked to serogroup Y strains of *Neisseria meningitidis*. Even a handful of cases triggers alarms because the disease spreads through respiratory droplets and close contact.
Epidemiologists at the CDC emphasize that outbreaks often appear in dense social networks—college dormitories, shelters, or large gatherings. Roughly 10% of people carry the bacteria in their throat without symptoms, creating silent reservoirs.
When cases appear, health authorities move quickly with antibiotics and targeted vaccination. That urgency reflects how rapidly symptoms can emerge once infection begins.
Q4: What are the earliest symptoms and why do doctors sometimes miss them?
Early meningitis symptoms mimic ordinary viral illness, making diagnosis challenging during the critical first hours. Patients typically develop fever, headache, nausea, and fatigue. Only later do classic neurological signs appear: neck stiffness, light sensitivity, confusion, or seizures. A landmark clinical analysis by Keith Cartwright at the University of Oxford showed meningococcal disease can progress from mild symptoms to septic shock within 24 hours.
Infants present an additional diagnostic puzzle. They cannot report headaches, so physicians rely on subtler clues such as irritability or a bulging fontanelle—the soft spot on a baby’s skull. Because symptoms evolve quickly, physicians rely on aggressive diagnostic tests to confirm infection.
Q5: How do doctors diagnose and treat meningitis today?
Doctors confirm meningitis by analyzing cerebrospinal fluid obtained through a lumbar puncture. During this procedure, clinicians withdraw a small CSF sample from the lower spine. In bacterial meningitis the fluid contains elevated white blood cells and proteins. Modern polymerase chain reaction testing—an amplification method that detects microbial DNA—can identify pathogens within hours, according to a 2020 review in *Nature Reviews Neurology*.
Treatment begins immediately with intravenous antibiotics such as ceftriaxone and anti-inflammatory drugs like dexamethasone. Vaccines have already transformed prevention: childhood immunization nearly eliminated *Haemophilus influenzae* type b meningitis in many countries. Yet scientists still debate why some infections trigger catastrophic inflammation while others remain mild.
Q6: How do meningitis vaccines actually work, and are they enough?
Meningitis vaccines are among the most mechanistically elegant tools in modern immunology — and also frustratingly incomplete. The short answer: they train the immune system to recognize the polysaccharide “coats” of specific bacterial strains before the brain ever encounters them.
Current meningococcal vaccines cover serogroups A, C, W, Y, and — as of the MenB vaccines licensed after 2014 — serogroup B, which historically caused roughly 30% of cases in Europe and North America and resisted earlier vaccine approaches. The reason group B was so hard to vaccinate against is genuinely fascinating: its polysaccharide capsule is chemically identical to a molecule found in human fetal neural tissue. Targeting it directly would trigger an autoimmune attack on the brain. Researchers at the Novartis Vaccines Institute (now GSK) circumvented this by switching strategy entirely — targeting surface proteins instead of the capsule, using a technique called reverse vaccinology pioneered by Rino Rappuoli.
The global picture, however, remains uneven. The MenAfriVac campaign, launched in 2010 across Africa’s “meningitis belt” — a swath of 26 countries from Senegal to Ethiopia — reduced serogroup A meningitis by over 99% in vaccinated populations by 2016. Yet serogroup C surged to partially fill the void, a phenomenon epidemiologists call “serotype replacement,” a challenge that remains unresolved.
No single vaccine covers all causative organisms — viral, fungal, and tubercular meningitis still have no broadly effective preventive tools.
Q7: What does cutting-edge research say about treating meningitis beyond antibiotics?
The antibiotic era saved millions, but researchers now recognize that killing the bacteria is only half the battle. The other half — suppressing the inflammatory cascade that the dying bacteria trigger — may be where future therapies live.
Bacterial lysis (the rupturing of bacterial cells by antibiotics) releases massive quantities of LPS and DNA fragments, which paradoxically intensify the very cytokine storm driving brain damage. This has led researchers at the University of Bern, particularly the group of Professor Stephen Leib, to investigate “anti-inflammatory adjunctive therapies” — treatments given alongside antibiotics specifically to dampen immune overreaction.
Daptomycin, an antibiotic that kills bacteria without causing lysis, showed significantly reduced brain inflammation compared to standard ceftriaxone in experimental models published in Journal of Infectious Diseases in 2018. It has not yet entered routine clinical use for meningitis, but trials are ongoing. Separately, mesenchymal stem cell therapy demonstrated neuroprotective effects in a 2023 rodent study at Sichuan University, reducing hippocampal (memory-center) neuron death by 60% post-infection.
The horizon is promising — but the translational gap between mouse models and human ICU patients remains enormous and honest researchers acknowledge it openly.
