Could a 1950s Epilepsy Medication Become a Breakthrough Treatment for Sleep Apnea?
Every night, nearly one billion people go to sleep and unknowingly stop breathing — again and again. Their airways collapse. Oxygen levels fall. The brain sends an emergency signal, forcing the body to briefly wake up just long enough to gasp for air before drifting back to sleep. By morning, they wake exhausted without knowing why.
For decades, the primary medical solution has been a machine: continuous positive airway pressure (CPAP) — a mask that delivers pressurized air to keep the airway open during sleep.But now, an unexpected candidate has emerged: a pill originally developed in the 1950s to treat epilepsy.
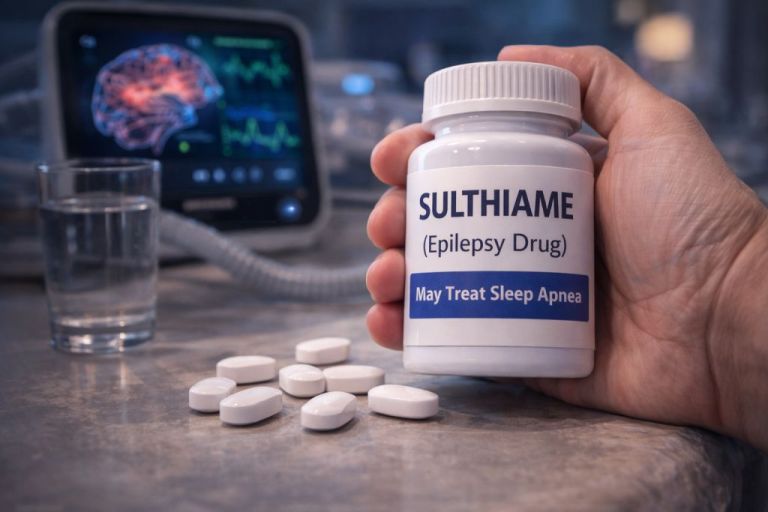
The drug is called sulthiame, and new research suggests it may dramatically reduce the breathing interruptions that define obstructive sleep apnea.
The Hidden Global Crisis: Obstructive Sleep Apnea
What is Obstructive Sleep Apnea (OSA)?
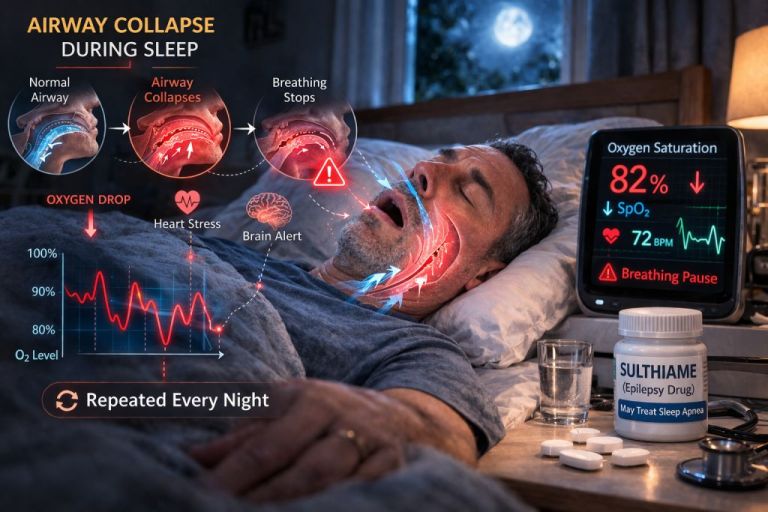
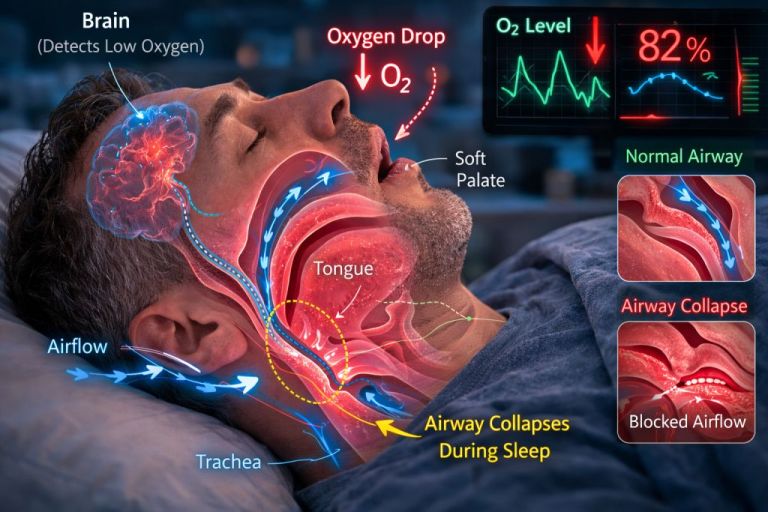
Obstructive sleep apnea occurs when the muscles of the upper airway relax too much during sleep, causing the throat to collapse.
Think of it like a garden hose being squeezed shut — airflow suddenly stops.
What happens during an apnea episode?
- The airway collapses.
- Oxygen levels drop.
- The brain senses danger.
- The sleeper partially wakes up to resume breathing.
This cycle may repeat dozens or even hundreds of times every night.
Why Sleep Apnea Is More Dangerous Than Most People Realize?
OSA does far more than cause poor sleep.
Long-term consequences include:
- Hypertension
- Cardiovascular disease
- Stroke
- Type 2 diabetes
- Cognitive decline
- Chronic fatigue
- Increased accident risk due to daytime sleepiness
Despite affecting an estimated one billion people worldwide, treatments remain limited.
The Limits of CPAP Therapy
The Current Gold Standard: CPAP
Continuous Positive Airway Pressure (CPAP) therapy works by delivering constant airflow through a mask that keeps the airway open.
When used correctly, CPAP is highly effective.
However, it has a major limitation:
Many patients simply cannot tolerate it.
Common complaints include:
- Mask discomfort
- Air leaks and noise
- Disrupted sleep
- Difficulty sleeping with a device attached to the face
- Reduced intimacy with partners
Studies show that up to 50% of patients abandon CPAP within one year.
For those who cannot tolerate CPAP, treatment options are extremely limited.

An Unexpected Candidate: Sulthiame
A Drug from the 1950s:
Sulthiame was first synthesized in the late 1950s and has long been prescribed for childhood epilepsy.
It is marketed under the brand name Ospolot in several regions, including:
- Europe
- Israel
- Japan
- Australia
Interestingly, the drug has never been approved in the United States.
How Sulthiame Works?
Sulthiame functions by inhibiting an enzyme called carbonic anhydrase.
What does carbonic anhydrase do?
This enzyme helps regulate the body’s breathing control system, influencing how the brain responds to changes in oxygen and carbon dioxide.
You can think of it as part of the body’s respiratory thermostat.
When sulthiame blocks this enzyme:
- The brain becomes more sensitive to rising carbon dioxide
- The drive to breathe increases
- Breathing becomes more stable during sleep
In simple terms, the brain becomes less willing to tolerate pauses in breathing.
The Scientific Breakthrough: The FLOW Trial
Who Led the Research?
The research was led by Jan Hedner, a senior professor of pulmonary medicine at the Sahlgrenska Academy, University of Gothenburg in Sweden.
Hedner asked a question many researchers had overlooked:
Could a seizure drug stabilize breathing during sleep?
To answer this, researchers conducted the FLOW trial, published in The Lancet (March 2026).
Key Details of the FLOW Trial
Study Design
- 298 adults with moderate to severe sleep apnea
- Conducted across five European countries
- Randomized, double-blind, placebo-controlled trial
- Participants took sulthiame one hour before bedtime
- Duration: 15 weeks
Participants were assigned to:
- Placebo
- Low dose sulthiame
- Moderate dose sulthiame
- High dose sulthiame
What the Study Found
The results were striking.
Major Outcomes
Participants receiving the highest dose experienced:
- Nearly 50% reduction in breathing interruptions
- Significant improvements in oxygen saturation
- Reduced daytime sleepiness
Researchers measured this using the AHI4 index, which tracks apnea events that cause oxygen drops.
Key Observation
The benefits were dose-dependent — higher doses produced stronger improvements.
Why Sulthiame Is Different from Other Treatments?
Most drug approaches to sleep apnea focus on the airway muscles.
Their goal is to physically prevent the throat from collapsing.
Sulthiame takes a completely different approach.
Instead of targeting the airway directly, it acts on:
- The brainstem respiratory control centers
- The body’s breathing regulation system
Think of the difference like this:
Traditional Treatments | Sulthiame |
Reinforce the airway | Stabilize breathing control |
Act locally in throat muscles | Act centrally in the brain |
Focus on structural support | Improve respiratory drive |
Rather than forcing the airway open, the drug reduces the likelihood that collapse happens at all.
Side Effects and Safety
The FLOW trial reported mostly mild side effects, including:
- Tingling sensations
- Mild headache
- Fatigue
These effects were:
- Dose-dependent
- Generally temporary
Researchers identified 200 mg per night as a likely optimal dose that balances effectiveness with tolerability.
Importantly: No serious safety concerns were detected during the study.
Why Researchers Are Still Cautious?
Despite promising results, experts stress that the findings are not yet definitive.
Sleep specialist Vishesh Kapur of the University of Washington highlighted several limitations:
Key Study Limitations
- Trial duration was only 15 weeks
- Women represented only 26% of participants
- Long-term safety data is still lacking
This is important because sleep apnea often presents differently in women and is frequently underdiagnosed. Larger Phase III trials will be required before regulatory approval.
Other Treatments Emerging for Sleep Apnea
The sleep apnea treatment pipeline is suddenly becoming more active.
New and emerging therapies include:
AD109
- Being developed by Apnimed
- Has already shown positive Phase III results
Tirzepatide
- A GLP-1 drug developed by Eli Lilly
- Approved by the FDA in 2024 for OSA
- Works indirectly through weight loss
Sulthiame may offer a different advantage.
Key difference
Sulthiame appears to work independently of body weight, which is important because many OSA patients are not obese.
The Ethical Question
The story of sulthiame raises a deeper question.
The drug has existed for more than 60 years.
It has been used safely for epilepsy in several countries.
Yet its potential role in treating sleep apnea — a disease affecting one billion people — remained largely unexplored.
If the drug ultimately proves effective, it means a potential therapy was sitting in plain sight for decades.
The cost of that delay could be enormous.
Untreated sleep apnea contributes to:
- Cardiovascular disease
- Metabolic disorders
- Cognitive impairment
- Reduced quality of life
Every year without better treatments represents millions of people living with preventable harm.
What Happens Next?
The FLOW trial provides strong proof of concept.
But several steps remain before sulthiame could become a widely available treatment.
Next steps include:
- Large Phase III clinical trials
- Regulatory review by agencies such as the FDA
- Pharmaceutical licensing and commercialization
- Integration into clinical practice
The science has now opened the door. What happens next will depend on the institutions responsible for translating discovery into treatment.
And those timelines rarely move as fast as the needs of a billion disrupted nights of sleep.
Scientific Reference
Randerath W. et al.
“Sultiame once per day in obstructive sleep apnoea (FLOW): a multicentre, randomised, double-blind, placebo-controlled, dose-finding, phase 2 trial.”
The Lancet, 2026
DOI: 10.1016/S0140-6736(25)01196-1

