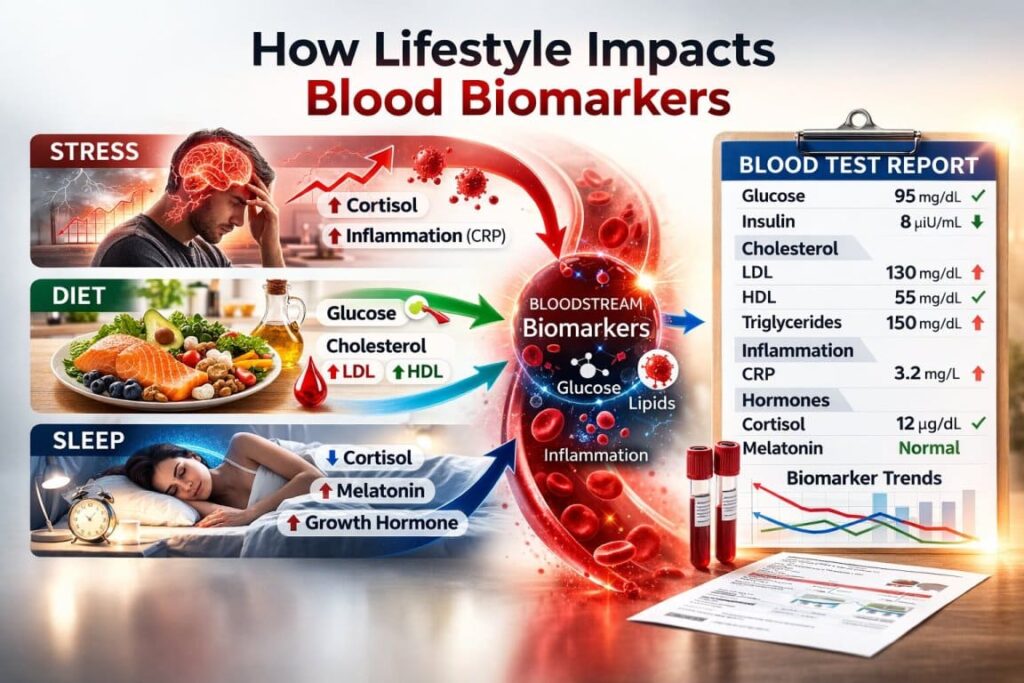
How Diet, Sleep & Stress Affect Blood Biomarkers?
Most people treat blood tests like a verdict. You get your report. You look at the numbers. You feel relieved — or worried. But here’s what’s rarely explained:
Your blood biomarkers are not fixed. They are constantly being rewritten. What you ate yesterday. How well you slept last week.The stress you’ve been carrying for months. All of it leaves a measurable chemical signature in your bloodstream.
This article explores — in depth — how diet, sleep, and stress act as biological programmers, shaping glucose control, inflammation, hormones, lipids, and long-term disease risk.
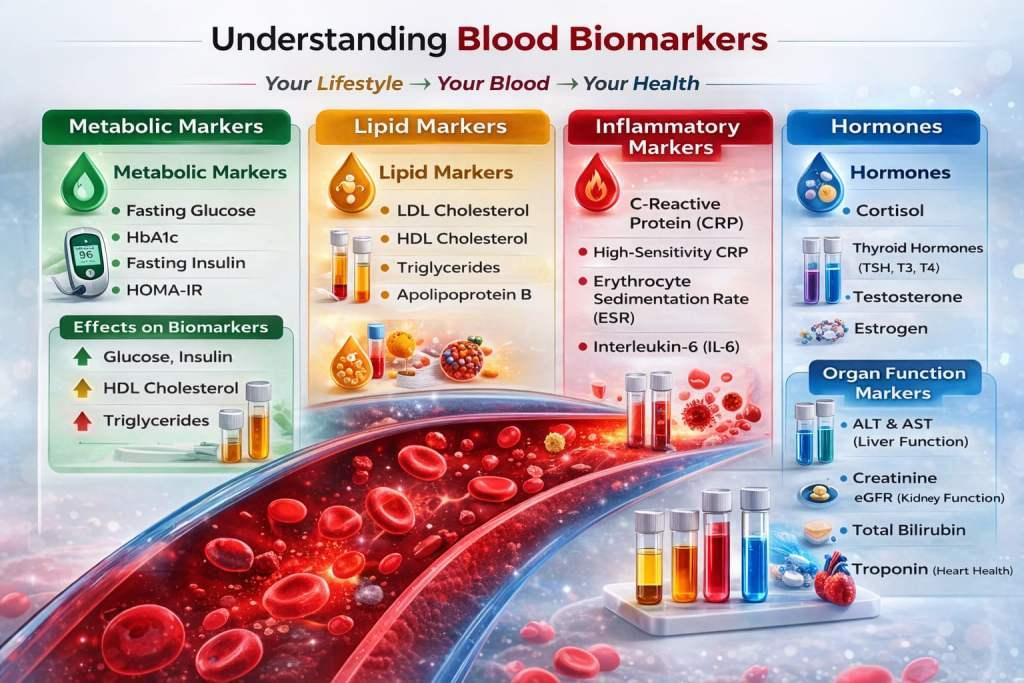
What Are Blood Biomarkers, Really?
Blood biomarkers are measurable substances that reflect biological processes inside your body. They give insight into:
- Metabolic health (glucose, insulin, HbA1c)
- Lipid metabolism (LDL, HDL, triglycerides, ApoB)
- Inflammation (CRP, hs-CRP, IL-6, TNF-α)
- Hormonal balance (cortisol, thyroid hormones, testosterone)
- Immune function (white blood cells, neutrophil-to-lymphocyte ratio)
- Organ performance (ALT, AST, creatinine, eGFR)
Think of them as real-time data from your internal environment.
They often change long before symptoms appear.
1. Diet: The Metabolic Blueprint
Diet doesn’t just influence weight.
It reshapes metabolic chemistry at a cellular level.
Glucose, Insulin & Metabolic Flexibility
Key biomarkers:
- Fasting glucose
- HbA1c
- Fasting insulin
- HOMA-IR
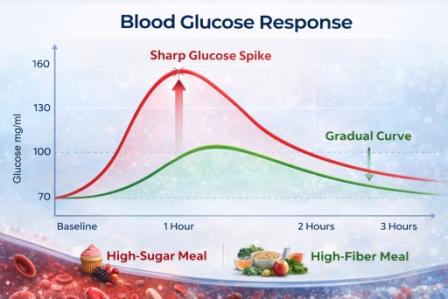
A high intake of refined carbohydrates and sugars triggers repeated glucose spikes. Over time:
- Insulin levels rise
- Cells become less responsive
- HbA1c increases (reflecting 3-month average glucose)
- Triglycerides rise through de novo lipogenesis (conversion of excess carbs into fat)
HbA1c is especially important. It measures “glycation” — when sugar binds to proteins, including hemoglobin. Think of it as biological “caramelization.”
On the other hand, diets rich in:
- Fiber
- Whole foods
- Legumes
- Vegetables
- Healthy fats
improve insulin sensitivity and stabilize glucose variability.
The triglyceride-to-HDL ratio is particularly revealing. A ratio above ~2.0 often signals insulin resistance more clearly than LDL alone.
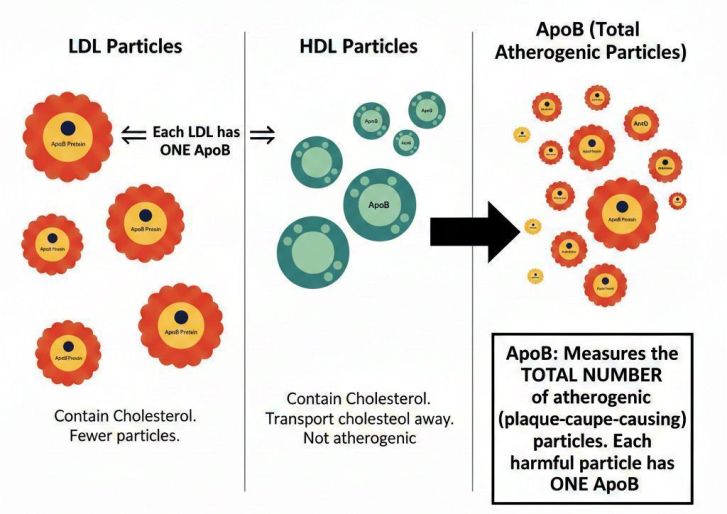
Lipid Profile: It’s Not Just LDL
Traditional focus stays on LDL cholesterol. But deeper analysis includes:
- Triglycerides
- HDL cholesterol
- ApoB (Apolipoprotein B)
ApoB measures the total number of atherogenic particles — a more precise cardiovascular risk marker than LDL concentration alone.
Refined sugars and trans fats can:
- Raise triglycerides
- Increase small dense LDL particles
- Lower HDL
Omega-3 fatty acids and Mediterranean-style eating patterns tend to:
- Reduce triglycerides
- Improve HDL function
- Lower inflammatory markers like CRP
Diet quality matters more than single nutrients.
Lipid Profile: It’s Not Just LDL
Traditional focus stays on LDL cholesterol. But deeper analysis includes:
- Triglycerides
- HDL cholesterol
- ApoB (Apolipoprotein B)
ApoB measures the total number of Atherogenic Particles — a more precise cardiovascular risk marker than LDL concentration alone.
Refined sugars and trans fats can:
- Raise triglycerides
- Increase small dense LDL particles
- Lower HDL
Omega-3 fatty acids and Mediterranean-style eating patterns tend to:
- Reduce triglycerides
- Improve HDL function
- Lower inflammatory markers like CRP
Diet quality matters more than single nutrients.
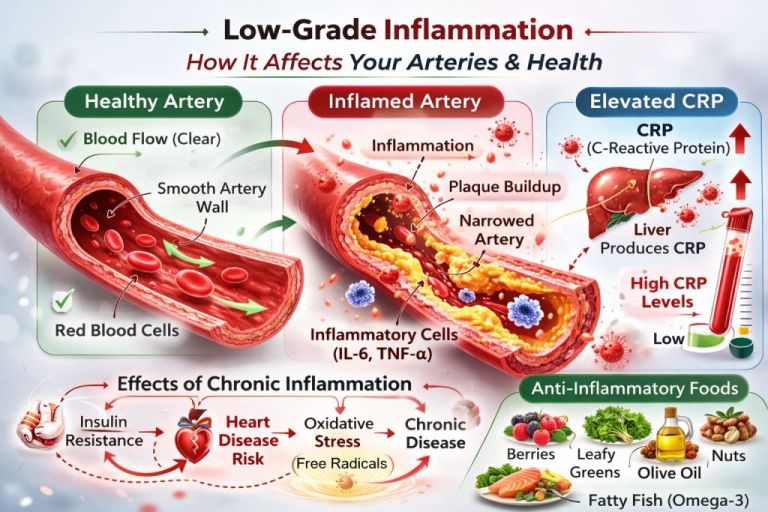
Inflammation: The Silent Chemical Fire
Chronic inflammation is like a low-grade internal fire — not dramatic like an infection, but persistent and damaging over time. When ultra-processed foods (high in refined sugars, seed oils, trans fats, additives, and low fiber) become a daily pattern, they continuously stimulate the immune system.
Over time, this can elevate key inflammatory biomarkers:
- CRP (C-Reactive Protein) – A general marker of systemic inflammation produced by the liver.
- hs-CRP (High-Sensitivity CRP) – A more precise version used to assess cardiovascular risk. Even mildly elevated levels can signal higher heart disease risk.
- IL-6 (Interleukin-6) – A signaling molecule (cytokine) that promotes inflammatory responses and is linked with metabolic dysfunction.
- TNF-α (Tumor Necrosis Factor-alpha) – A powerful inflammatory cytokine associated with insulin resistance and chronic disease progression.
Why This Matters?
Inflammation is not just something that happens during infections or injuries. It can quietly simmer in the background due to:
- Repeated blood sugar spikes
- Excess visceral fat
- Poor sleep patterns
- Chronic psychological stress
- Highly processed dietary patterns
This low-grade inflammation contributes to:
- Insulin resistance
- Atherosclerosis (plaque buildup in arteries)
- Fatty liver
- Hormonal imbalance
- Accelerated aging
The Oxidative Stress Connection
Ultra-processed foods increase oxidative stress — an imbalance between free radicals and the body’s antioxidant defenses. Oxidative stress activates inflammatory pathways, creating a feedback loop:
Poor diet → Oxidative stress → Cytokine release → More inflammation
How to Cool the Fire?
Anti-inflammatory, antioxidant-rich foods help interrupt this cycle:
- Berries (anthocyanins reduce oxidative damage)
- Leafy greens (rich in magnesium and polyphenols)
- Extra virgin olive oil (contains oleocanthal, a natural anti-inflammatory compound)
- Nuts and seeds (provide healthy fats and vitamin E)
- Fatty fish (omega-3 fatty acids lower inflammatory signaling)
Instead of eliminating inflammation entirely (which is impossible and unhealthy), the goal is regulation — keeping the immune system responsive but not chronically overactivated.
In simple terms:
Acute inflammation protects you.
Chronic inflammation slowly harms you.
Micronutrients & Oxidative Stress
Blood levels of:
- Vitamin D
- B12
- Iron
- Magnesium
- Folate
Often reflect dietary patterns before symptoms appear. Low antioxidant intake may increase oxidative stress biomarkers, accelerating cellular aging.
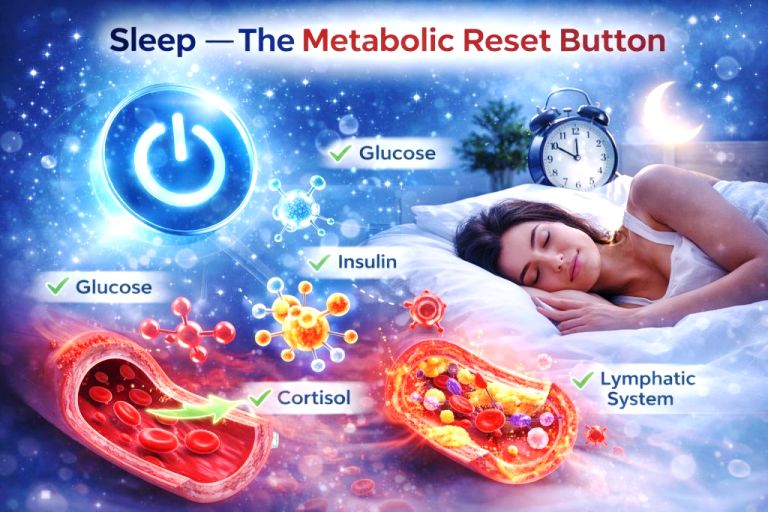
2. Sleep: The Metabolic Reset Button
Sleep is not downtime.
It is when your body recalibrates hormones, clears metabolic waste, and restores immune balance.
Even one night of partial sleep deprivation can induce temporary insulin resistance in healthy individuals.
Sleep & Glucose Control
Short sleep duration:
- Raises fasting glucose
- Increases cortisol
- Elevates fasting insulin
Sleep loss reduces insulin sensitivity independent of diet.
In other words:
You can eat perfectly — and still see glucose rise if you chronically under-sleep.
Sleep & Inflammation
Consistently sleeping under 6 hours is associated with:
- Elevated hs-CRP
- Increased inflammatory cytokines
- Higher white blood cell counts
Deep sleep (Stage 3) plays a role in clearing metabolic waste via the glymphatic system. When deep sleep is disrupted, inflammatory markers often rise the next day.
Sleep & Hormonal Balance
Sleep regulates:
- Cortisol (should peak in morning, decline at night)
- Growth hormone (released during deep sleep)
- Testosterone
- Melatonin
- Thyroid hormone regulation
Chronic sleep restriction can flatten cortisol rhythms — leading to the classic “wired but tired” feeling.
Sleep & Lipid Metabolism
Circadian disruption affects cholesterol-processing genes. Research suggests inadequate sleep may:
- Increase triglycerides
- Decrease HDL
- Impair lipid oxidation
Sleep timing matters as much as sleep duration.
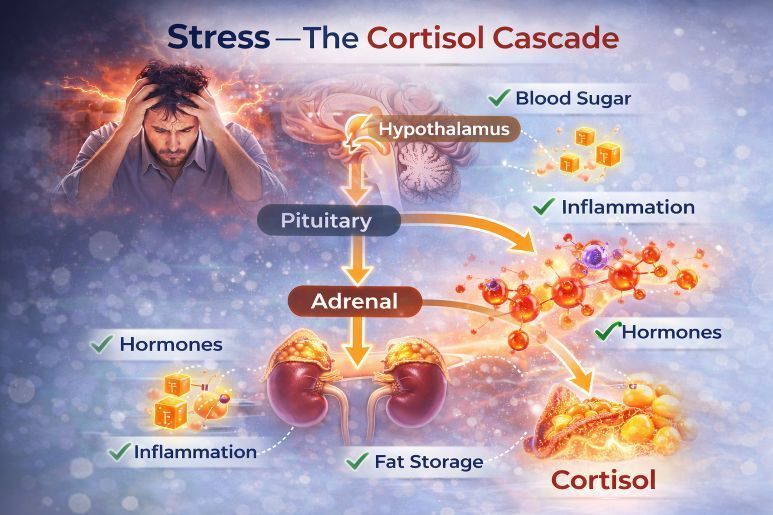
3. Stress: The Cortisol Cascade
Stress is not just emotional — it is biochemical.
When activated repeatedly, the HPA (hypothalamic-pituitary-adrenal) axis alters glucose metabolism, immune function, and cardiovascular risk markers.
Cortisol & Blood Sugar
Chronic stress elevates cortisol and adrenaline, which:
- Increase gluconeogenesis
- Raise fasting glucose
- Promote insulin resistance
- Encourage visceral fat storage
This is why some individuals with “healthy diets” still show elevated glucose.
Stress & Inflammation
Long-term stress increases:
- CRP
- IL-6
- TNF-α
- Fibrinogen
It also shifts immune balance.
The Neutrophil-to-Lymphocyte Ratio (NLR) is an emerging stress biomarker. Chronic stress raises neutrophils (innate defense) and lowers lymphocytes (adaptive immunity), increasing infection vulnerability.
Stress & Cardiovascular Risk
Stress-related biomarker shifts include:
- Elevated fibrinogen (clotting risk)
- Increased LDL oxidation
- Higher blood pressure
Over time, this contributes to cardiovascular disease risk.
Case Study Thinking: Treating the System, Not Just the Number
Imagine elevated fasting glucose.
A conventional response might be medication.
A deeper analysis asks:
- Is it dietary excess?
- Is it cortisol from stress?
- Is it sleep deprivation impairing insulin sensitivity?
Without examining all three, you risk treating the symptom rather than the underlying driver.
1. Dietary Excess (The Fuel Input)
This is the most direct lever, but it’s often about more than just “eating too much sugar.”
- The Mechanism: Constant intake of refined carbohydrates and ultra-processed foods keeps insulin levels chronically high. Over time, the body’s cells become “deaf” to insulin (insulin resistance).
- The System View: High glucose here is a capacity issue. The body’s storage tanks (muscle and liver glycogen) are full, so the excess “spills over” into the bloodstream. Treating this with medication without changing the fuel input is like trying to bail water out of a boat without plugging the leak.
2. Cortisol from Stress (The Internal Release)
You can eat a “perfect” diet and still have high blood sugar if your nervous system is in a state of chronic “fight or flight.”
- The Mechanism: Cortisol is a glucocorticoid. Its job is to mobilize energy. It signals the liver to dump stored glucose into the blood so you have the energy to “fight” a perceived threat.
- The System View: High glucose here is a signaling issue. If you are chronically stressed, your liver is constantly “printing money” (glucose) that you aren’t spending. Medication might lower the sugar, but it won’t stop the stress hormones from continuing to strain your cardiovascular system.
3. Sleep Deprivation (The Regulatory Failure)
Sleep is the “reboot” phase for metabolic hormones. Even a single night of poor sleep can induce a temporary state of pre-diabetes.
- The Mechanism: Sleep deprivation increases evening cortisol and decreases insulin sensitivity. It also disrupts “hunger hormones” (leptin and ghrelin), making you crave the very foods that worsen the glucose spike.
- The System View: High glucose here is a maintenance issue. The body hasn’t had the downtime required to clear metabolic waste and reset its hormonal sensitivity.
Why the Distinction Matters
Feature | Conventional Response | Systems-Based Response |
Focus | The Glucose Number | The Biological Context |
Goal | Get the lab value into the “green” range. | Identify which pillar of health is sagging. |
Risk | Side effects and lifelong dependence. | Misdiagnosis of the root cause. |
Outcome | Controlled symptoms. | Reclaimed metabolic flexibility. |
The Takeaway: If you treat high glucose caused by sleep deprivation with a pill, you might “fix” the blood work, but the patient remains exhausted, inflamed, and metabolically fragile.

Final Perspective: Your Blood Is a Feedback System
Your biomarkers are not destiny. They are signals.
They respond to:
- What you eat
- How you sleep
- How you handle stress
Over months and years, these inputs shape either metabolic resilience or chronic dysfunction. When you understand this, blood work becomes empowering — not frightening.
Your habits write the chemistry. Your chemistry shapes your future.
Scientific References & Sources
The information presented in this article is based on peer-reviewed research, large clinical reviews, and established medical guidelines from reputable institutions.
Diet & Blood Biomarkers
Triglycerides, HDL & Insulin Resistance
McLaughlin T. et al. “Use of Metabolic Markers to Identify Overweight Individuals Who Are Insulin Resistant.” Annals of Internal Medicine.
https://www.acpjournals.org/doi/full/10.7326/0003-4819-139-10-200311180-00007
Apolipoprotein B (ApoB) & Cardiovascular Risk
Sniderman A. et al. “Apolipoprotein B versus Non–HDL Cholesterol.” Journal of the American College of Cardiology (JACC).
Mediterranean Diet & Cardiovascular Risk
Estruch R. et al. “Primary Prevention of Cardiovascular Disease with a Mediterranean Diet.” New England Journal of Medicine (NEJM).
Diet, Inflammation & CRP
Calder PC. et al. “Dietary factors and low-grade inflammation.” British Journal of Nutrition.
Sleep & Blood Biomarkers
Sleep Restriction & Insulin Sensitivity
Spiegel K. et al. “Impact of Sleep Debt on Metabolic and Endocrine Function.” The Lancet.
https://www.thelancet.com/journals/lancet/article/PIIS0140673601086906
Sleep Duration & Inflammation (CRP, IL-6)
Irwin MR. “Sleep and Inflammation.” Sleep.
Sleep & Glycemic Control
Reutrakul S. & Van Cauter E. “Sleep Influences on Obesity, Insulin Resistance, and Risk of Type 2 Diabetes.” Metabolism.
https://www.sciencedirect.com/science/article/pii/S0026049513002084
Sleep & Hormonal Regulation
Leproult R. & Van Cauter E. “Role of Sleep in the Regulation of Hormones.” Endocrine Development.
Stress & Blood Biomarkers
Stress, Cortisol & Glucose Metabolism
Tsigos C. & Chrousos GP. “Hypothalamic–Pituitary–Adrenal Axis and Stress.” Journal of Psychosomatic Research.
https://www.sciencedirect.com/science/article/pii/S0022399902005487
Psychological Stress & Inflammation (IL-6, TNF-α)
Black PH. & Garbutt LD. “Stress, Inflammation and Cardiovascular Disease.” Journal of Psychosomatic Research.
Neutrophil-to-Lymphocyte Ratio as Stress Marker
Zahorec R. “Neutrophil-to-Lymphocyte Ratio.” Bratislavske Lekarske Listy.
Chronic Stress & Cardiovascular Risk
Steptoe A. & Kivimäki M. “Stress and Cardiovascular Disease.” Nature Reviews Cardiology.
General Medical References
American Heart Association (AHA)
Cholesterol & Cardiovascular Risk Guidelines
Centers for Disease Control and Prevention (CDC)
Biomarkers & Chronic Disease Data
National Institutes of Health (NIH)
Sleep, Stress & Metabolic Health

